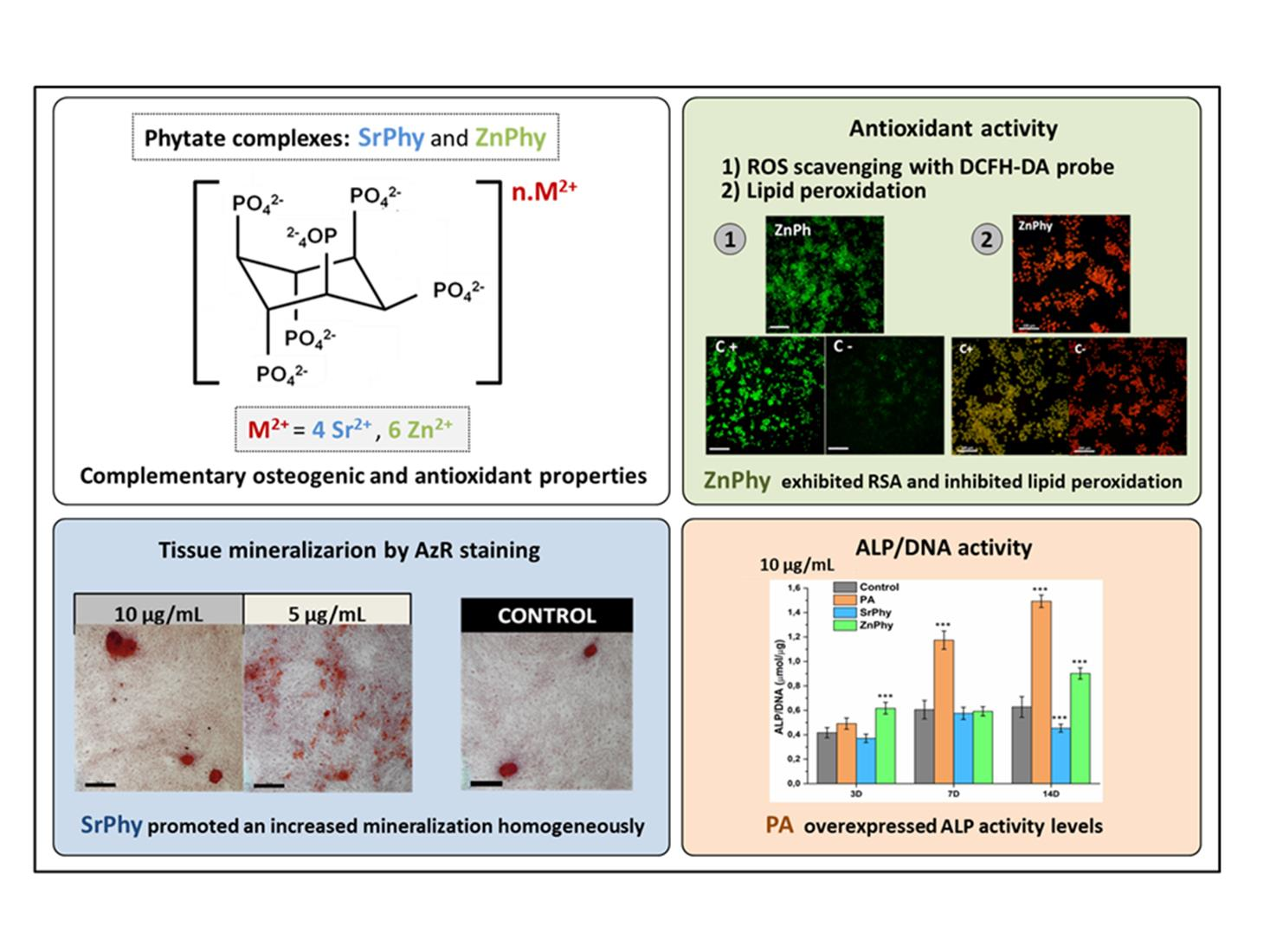
Sr/Zn phytate compounds have been shown interest in biomaterial science, specifically in dental implantology, due to their antimicrobial effects against Streptococcus mutans and their capacity to form bioactive coatings. Phytic acid is a natural chelating compound that shows antioxidant and osteogenic properties that can play an important role in bone remodelling processes affected by oxidative stress environments, such as those produced during infections. The application of non-protein cell-signalling molecules that regulate both bone and ROS homeostasis is a promising strategy for the regeneration of bone tissues affected by oxidative stress processes. In this context, phytic acid (PA) emerged as an excellent option since its antioxidant and osteogenic properties can play an important role in bone remodelling processes. In this study, we explored the antioxidant and osteogenic properties of two metallic PA complexes bearing bioactive cations, i.e., Sr2+ (SrPhy) and Zn2+ (ZnPhy), highlighting the effect of the divalent cations anchored to phytate moieties and their capability to modulate the PA properties. The in vitro features of the complexes were analyzed and compared with those of their precursor PA. The ferrozine/FeCl2 method indicated that SrPhy exhibited a more remarkable ferrous ion affinity than ZnPhy, while the antioxidant activity demonstrated by a DPPH assay showed that only ZnPhy reduced the content of free radicals. Likewise, the antioxidant potential was assessed with RAW264.7 cell cultures. An ROS assay indicated again that ZnPhy was the only one to reduce the ROS content (20%), whereas all phytate compounds inhibited lipid peroxidation following the decreasing order of PA > SrPhy > ZnPhy. The in vitro evaluation of the phytate’s osteogenic ability was performed using hMSC cells. The results showed tailored properties related to the cation bound in each complex. ZnPhy overexpressed ALP activity at 3 and 14 days, and SrPhy significantly increased calcium deposition after 21 days. This study demonstrated that Sr/Zn phytates maintained the antioxidant and osteogenic properties of PA and can be used in bone regenerative therapies involving oxidative environments, such as infected implant coatings and periodontal tissues.
Strontium/zinc phytate-based self-assembled monolayers on titanium surfaces enhance osteogenesis and antibacterial performance in vitro
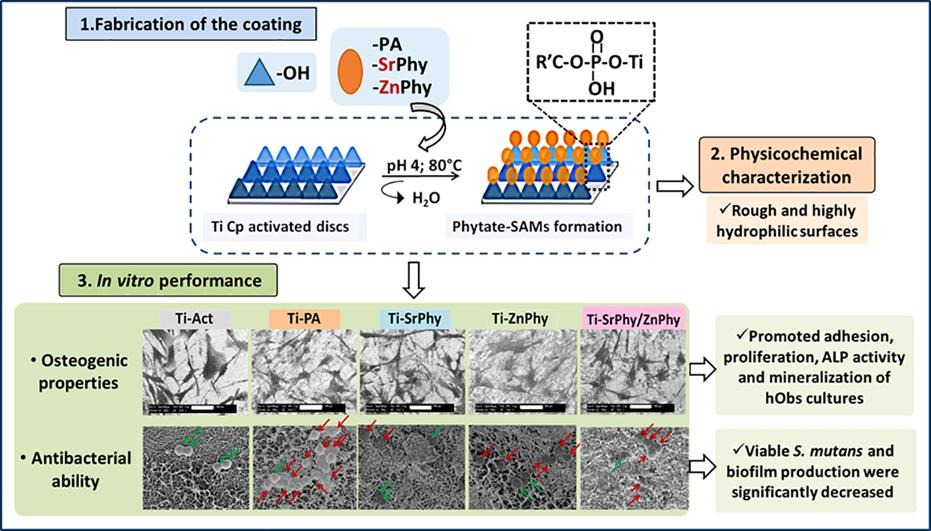
The accumulation of bacteria over implant surfaces is still the first cause of failure, and the development of antimicrobial surfaces constitutes a first line in implant research. Besides, the durability and mechanical performance of implants, in special in the dental area, are mainly determined by their osseointegration capacity into the maxillofacial bone and the appearance of infections. Consequently, implant osseointegration and infection prophylaxis remain as big challenges to attain so a huge investigation is being developed on the production of bioactive surfaces to achieve improvements in these aspects. In this work we propose the functionalization of titanium surfaces (Ti Cp) with self-assembled monolayers (SAMs) of bioactive organophosphate compounds: phytic acid (Ti-PA) and its metallic phytate derivatives bearing Sr2+ and/or Zn2+ (Ti-SrPhy, Ti-ZnPhy and Ti-SrPhy/ZnPhy) which exhibited tunable in vitro osteogenic, antimicrobial and antioxidant properties in a previous work. Thus, phytate compounds are chemically anchored onto Ti discs through a simple procedure consisting of a condensation reaction promoted by heat treatment. EDS and XPS spectroscopies confirm the obtaining of the modified surfaces and the topographic properties and wettability analysed by SEM, AFM, profilometry and contact angle measurements, respectively, are explored. Additionally, phytate-SAMs do not release any cytotoxic compound after 14 days and stimulate in vitro adhesion and proliferation of human osteoblast cells after 14 days of culture. The osteogenic ability of the modified surfaces evaluated by the quantification of ALP activity and matrix mineralization degree shows a significant improvement with respect to unmodified surfaces. Furthermore, the antimicrobial activity of phytate-SAMs against Streptococcus mutans cultures is evaluated. The count of viable cells and the quantification of produced biofilm are significantly reduced by all phytate-SAMs groups (p < 0.001). Cell membrane integrity studies by LIVE/DEAD staining and SEM imaging confirm a decreased viability of adhered bacteria when phytate-based surfaces are tested, due to a disruption in the function and permeability of the cell membrane. Therefore, phytate-SAMs exhibit suitable in vitro features suggesting their promising potential as bioactive coatings of dental implants.
A study on Sr/Zn phytate complexes: structural properties and antimicrobial synergistic effects against Streptococcus mutans
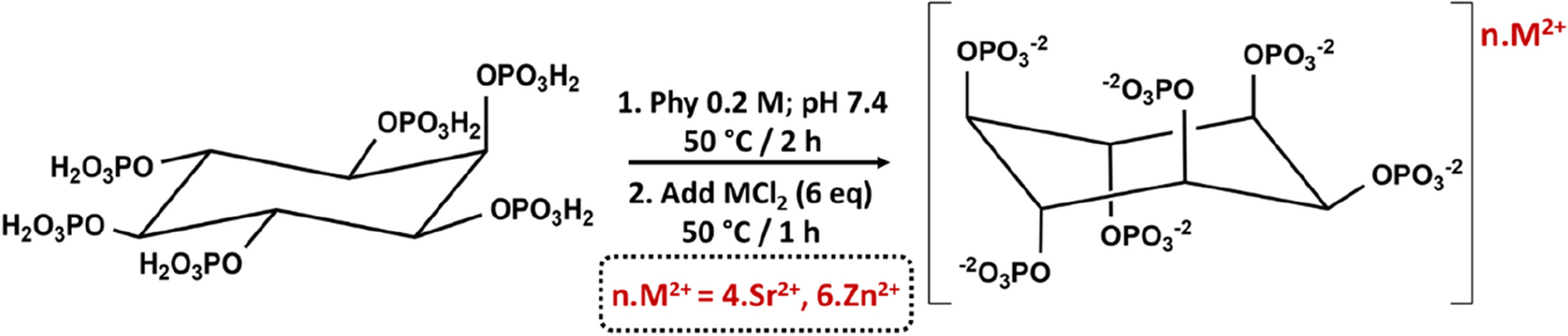
Phytic acid (PA) is an abundant natural plant component that exhibits a versatility of applications benefited from its chemical structure, standing out its use as food, packing and dental additive due to its antimicrobial properties. The capacity of PA to chelate ions is also well-established and the formation and thermodynamic properties of different metallic complexes has been described. However, research studies of these compounds in terms of chemistry and biological features are still demanded in order to extend the application scope of PA complexes. The main goal of this paper is to deepen in the knowledge of the bioactive metal complexes chemistry and their bactericide activity, to extend their application in biomaterial science, specifically in oral implantology. Thus, this work presents the synthesis and structural assessment of two metallic phytate complexes bearing the bioactive cations Zn2+ and Sr2+ (ZnPhy and SrPhy respectively), along with studies on the synergic biological properties between PA and cations. Metallic phytates were synthesized in the solid-state by hydrothermal reaction leading to pure solid compounds in high yields. Their molecular formulas were C6H12024P6Sr4·5H2O and C6H12024P6Zn6·6H2O, as determined by ICP and HRES-TGA. The metal coordination bond of the solid complexes was further analysed by EDS, Raman, ATR-FTIR and solid 13C and 31P-NMR spectroscopies. Likewise, we evaluated the in vitro ability of the phytate compounds for inhibiting biofilm production of Streptococcus mutans cultures. Results indicate that all compounds significantly reduced biofilm formation (PA < SrPhy < ZnPhy), and ZnPhy even showed remarkable differences with respect to PA and SrPhy. Analysis of antimicrobial properties shows the first clues of the possible synergic effects created between PA and the corresponding cation in different cell metabolic processes. In overall, findings of this work can contribute to expand the applications of these bioactive metallic complexes in the biotechnological and biomedical fields, and they can be considered for the fabrication of anti-plaque coating systems in the dentistry field.