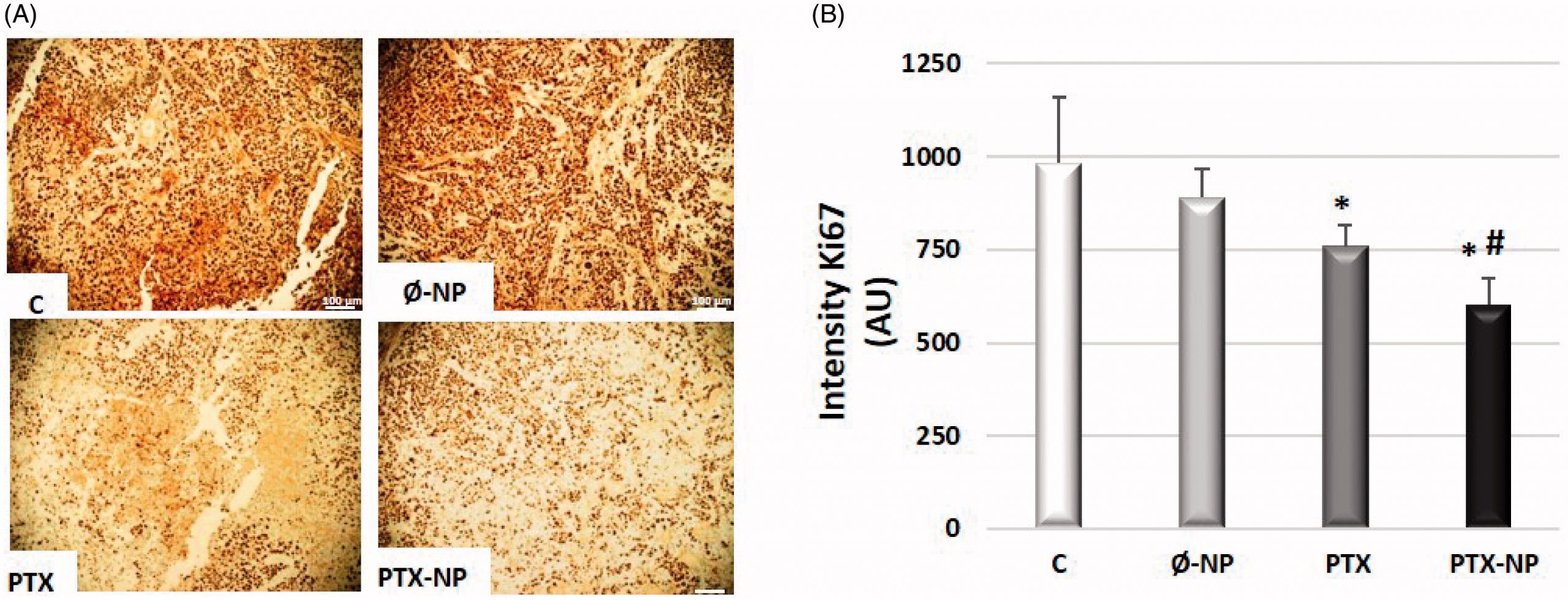
The prognosis of patients with recurrent or metastatic head and neck squamous cell cancer (HNSCC) is generally poor. New treatments are required to supplement the current standard of care. Paclitaxel (PTX), an effective chemotherapeutic for HNSCC, has serious side effects. A polymeric nanocarrier system was developed for the delivery of PTX to improve HNSCC treatment. This study aimed to evaluate the antitumor efficacy of PTX-loaded polymeric nanoparticles based on α-TOS (PTX-NPs) administered by direct intratumoral injection into a Hypopharynx carcinoma squamous cells (FaDu) tumor xenograft mouse model. The nanocarrier system based on block copolymers of polyethylene glycol (PEG) and a methacrylic derivative of α-TOS was synthesized and PTX was loaded into the delivery system. Tumor volume was measured to evaluate the antitumor effect of the PTX-NPs. The relative mechanisms of apoptosis, cell proliferation, growth, angiogenesis, and oxidative and nitrosative stress were detected by Western blotting, fluorescent probes, and immunohistochemical analysis. The antitumor activity results showed that compared to free PTX, PTX-NPs exhibited much higher antitumor efficacy and apoptosis-inducing in a FaDu mouse xenograft model and demonstrated an improved safety profile. Ki-67, EGFR, and angiogenesis markers (Factor VIII, CD31, and CD34) expression were significantly lower in the PTX-NPs group compared with other groups (p < .05). Also, PTX-NPs induced oxidative and nitrosative stress in tumor tissue. Direct administration of PTX-loaded polymeric nanoparticles based on α-Tocopheryl Succinate at the tumor sites, proved to be promising for HNSCC therapy.
3729048
{3729048:PKVECIFS}
nature
50
1
1
title
931
http://www.biomateriales.ictp.csic.es/wp-content/plugins/zotpress/
%7B%22status%22%3A%22success%22%2C%22updateneeded%22%3Afalse%2C%22instance%22%3A%22zotpress-355d526cfc536514d0366c4fd2c5a94e%22%2C%22meta%22%3A%7B%22request_last%22%3A0%2C%22request_next%22%3A0%2C%22used_cache%22%3Atrue%7D%2C%22data%22%3A%5B%7B%22key%22%3A%22PKVECIFS%22%2C%22library%22%3A%7B%22id%22%3A3729048%7D%2C%22meta%22%3A%7B%22creatorSummary%22%3A%22Riestra-Ayora%20et%20al.%22%2C%22parsedDate%22%3A%222021-01-01%22%2C%22numChildren%22%3A2%7D%2C%22bib%22%3A%22%3Cdiv%20class%3D%5C%22csl-bib-body%5C%22%20style%3D%5C%22line-height%3A%202%3B%20%5C%22%3E%5Cn%20%3Cdiv%20class%3D%5C%22csl-entry%5C%22%20style%3D%5C%22clear%3A%20left%3B%20%5C%22%3E%5Cn%20%3Cdiv%20class%3D%5C%22csl-left-margin%5C%22%20style%3D%5C%22float%3A%20left%3B%20padding-right%3A%200.5em%3B%20text-align%3A%20right%3B%20width%3A%201em%3B%5C%22%3E1.%3C%5C%2Fdiv%3E%3Cdiv%20class%3D%5C%22csl-right-inline%5C%22%20style%3D%5C%22margin%3A%200%20.4em%200%201.5em%3B%5C%22%3ERiestra-Ayora%2C%20J.%20%3Ci%3Eet%20al.%3C%5C%2Fi%3E%20%3Ca%20target%3D%27_blank%27%20href%3D%27https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1080%5C%2F10717544.2021.1923863%27%3EPaclitaxel-loaded%20polymeric%20nanoparticles%20based%20on%20%5Cu03b1-tocopheryl%20succinate%20for%20the%20treatment%20of%20head%20and%20neck%20squamous%20cell%20carcinoma%3A%20in%20vivo%20murine%20model%3C%5C%2Fa%3E.%20%3Ci%3EDrug%20Delivery%3C%5C%2Fi%3E%20%3Cb%3E28%3C%5C%2Fb%3E%2C%201376%5Cu20131388%20%282021%29.%20%3Ca%20title%3D%27Cite%20in%20RIS%20Format%27%20class%3D%27zp-CiteRIS%27%20href%3D%27http%3A%5C%2F%5C%2Fwww.biomateriales.ictp.csic.es%5C%2Fwp-content%5C%2Fplugins%5C%2Fzotpress%5C%2Flib%5C%2Frequest%5C%2Frequest.cite.php%3Fapi_user_id%3D3729048%26amp%3Bitem_key%3DPKVECIFS%27%3ECite%3C%5C%2Fa%3E%20%3C%5C%2Fdiv%3E%5Cn%20%3C%5C%2Fdiv%3E%5Cn%3C%5C%2Fdiv%3E%22%2C%22data%22%3A%7B%22itemType%22%3A%22journalArticle%22%2C%22title%22%3A%22Paclitaxel-loaded%20polymeric%20nanoparticles%20based%20on%20%5Cu03b1-tocopheryl%20succinate%20for%20the%20treatment%20of%20head%20and%20neck%20squamous%20cell%20carcinoma%3A%20in%20vivo%20murine%20model%22%2C%22creators%22%3A%5B%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Juan%22%2C%22lastName%22%3A%22Riestra-Ayora%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Carolina%22%2C%22lastName%22%3A%22S%5Cu00e1nchez-Rodr%5Cu00edguez%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Raquel%22%2C%22lastName%22%3A%22Palao-Suay%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Joaqu%5Cu00edn%22%2C%22lastName%22%3A%22Yanes-D%5Cu00edaz%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Ana%22%2C%22lastName%22%3A%22Mart%5Cu00edn-Hita%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Mar%5Cu00eda%20Rosa%22%2C%22lastName%22%3A%22Aguilar%22%7D%2C%7B%22creatorType%22%3A%22author%22%2C%22firstName%22%3A%22Ricardo%22%2C%22lastName%22%3A%22Sanz-Fern%5Cu00e1ndez%22%7D%5D%2C%22abstractNote%22%3A%22The%20prognosis%20of%20patients%20with%20recurrent%20or%20metastatic%20head%20and%20neck%20squamous%20cell%20cancer%20%28HNSCC%29%20is%20generally%20poor.%20New%20treatments%20are%20required%20to%20supplement%20the%20current%20standard%20of%20care.%20Paclitaxel%20%28PTX%29%2C%20an%20effective%20chemotherapeutic%20for%20HNSCC%2C%20has%20serious%20side%20effects.%20A%20polymeric%20nanocarrier%20system%20was%20developed%20for%20the%20delivery%20of%20PTX%20to%20improve%20HNSCC%20treatment.%20This%20study%20aimed%20to%20evaluate%20the%20antitumor%20efficacy%20of%20PTX-loaded%20polymeric%20nanoparticles%20based%20on%20%5Cu03b1-TOS%20%28PTX-NPs%29%20administered%20by%20direct%20intratumoral%20injection%20into%20a%20Hypopharynx%20carcinoma%20squamous%20cells%20%28FaDu%29%20tumor%20xenograft%20mouse%20model.%20The%20nanocarrier%20system%20based%20on%20block%20copolymers%20of%20polyethylene%20glycol%20%28PEG%29%20and%20a%20methacrylic%20derivative%20of%20%5Cu03b1-TOS%20was%20synthesized%20and%20PTX%20was%20loaded%20into%20the%20delivery%20system.%20Tumor%20volume%20was%20measured%20to%20evaluate%20the%20antitumor%20effect%20of%20the%20PTX-NPs.%20The%20relative%20mechanisms%20of%20apoptosis%2C%20cell%20proliferation%2C%20growth%2C%20angiogenesis%2C%20and%20oxidative%20and%20nitrosative%20stress%20were%20detected%20by%20Western%20blotting%2C%20fluorescent%20probes%2C%20and%20immunohistochemical%20analysis.%20The%20antitumor%20activity%20results%20showed%20that%20compared%20to%20free%20PTX%2C%20PTX-NPs%20exhibited%20much%20higher%20antitumor%20efficacy%20and%20apoptosis-inducing%20in%20a%20FaDu%20mouse%20xenograft%20model%20and%20demonstrated%20an%20improved%20safety%20profile.%20Ki-67%2C%20EGFR%2C%20and%20angiogenesis%20markers%20%28Factor%20VIII%2C%20CD31%2C%20and%20CD34%29%20expression%20were%20significantly%20lower%20in%20the%20PTX-NPs%20group%20compared%20with%20other%20groups%20%28p%20%3C%20.05%29.%20Also%2C%20PTX-NPs%20induced%20oxidative%20and%20nitrosative%20stress%20in%20tumor%20tissue.%20Direct%20administration%20of%20PTX-loaded%20polymeric%20nanoparticles%20based%20on%20%5Cu03b1-Tocopheryl%20Succinate%20at%20the%20tumor%20sites%2C%20proved%20to%20be%20promising%20for%20HNSCC%20therapy.%22%2C%22date%22%3A%22January%201%2C%202021%22%2C%22language%22%3A%22%22%2C%22DOI%22%3A%2210.1080%5C%2F10717544.2021.1923863%22%2C%22ISSN%22%3A%221071-7544%22%2C%22url%22%3A%22https%3A%5C%2F%5C%2Fdoi.org%5C%2F10.1080%5C%2F10717544.2021.1923863%22%2C%22collections%22%3A%5B%5D%2C%22dateModified%22%3A%222021-07-07T09%3A26%3A54Z%22%7D%7D%5D%7D